Fission uranium 235 formula reaction create se
Three types of moderators are used at the MIT reactor: (1) ordinary or "light" water that is also used to cool the reactor core, (2) deuterated or heavy water (D 20), and (3) high-purity graphite, both of which are excellent at slowing neutrons without absorbing them.

Since U-235 nuclei do not readily absorb the high energy neutrons that are emitted during fission, it is necessary to slow the neutrons down with a "moderator". In the MIT reactor, one other group of components is essential to the maintaining and controlling a chain reaction. As fewer and fewer neutrons are absorbed, more and more neutrons are available to cause the splitting of uranium nuclei, until finally enough neutrons are available to sustain a chain reaction. To put the reactor into operation, the control blades are raised very slowly. When the control blades are fully inserted, they absorb so many neutrons from the uranium that there are not enough to allow a chain reaction to continue. Boron has the property of absorbing neutrons without re-emitting any. If the reaction will sustain itself, it is said to be 'critical', and the mass of U-235 required to produced the critical condition is said to be a 'critical mass'.A critical chain reaction can be achieved at low concentrations of U-235 if the neutrons from. The rate of fissions in the uranium nuclei in the MIT reactor is controlled chiefly by six control blades of boron-stainless steel which are inserted vertically alongside the fuel elements. If an least one neutron from U-235 fission strikes another nucleus and causes it to fission, then the chain reaction will continue. We now look at the fission of a single U-235 atom in more detail. (Compare fission of 1 g of U-235 with burning 1 kg of coal). A single fission does not create much energy but a gram of natural uranium contains 1.8 x 1019 U-235 atoms, and a CANDU reactor can fission about of these. When it is in operation, the central active core contains a huge number of neutrons traveling in every direction at very high speeds. gram of U-235 would produce 1 MWd of thermal energy. The MIT Research Reactor is used primarily for the production of neutrons. Hence, the possibility exists for creating a chain reaction. Each time a U-235 nucleus splits, it releases two or three neutrons. This process is known as fission (see diagram below).

When a U-235 nucleus absorbs an extra neutron, it quickly breaks into two parts. The arrangement of particles within uranium-235 is somewhat unstable and the nucleus can disintegrate if it is excited by an outside source. The atomic numbers are already balanced, since you have 92 + 0 on the reactants' side, and 52 + 40 on the products' side.In the nucleus of each atom of uranium-235 (U-235) are 92 protons and 143 neutrons, for a total of 235.
Fission uranium 235 formula reaction create se free#
The fission process may produce 2, 3, or more free neutrons, and these neutrons can trigger further fission, and a chain reaction can occur. #"235 + 1" = 137 + 97 + color(red)(?)*1 => color(red)(?) = 236 - 234 = 2# Some nuclei can undergo fission on their own spontaneously, but only specific nuclei, like uranium-235, uranium-233, and plutonium-239, can sustain a fission chain reaction.
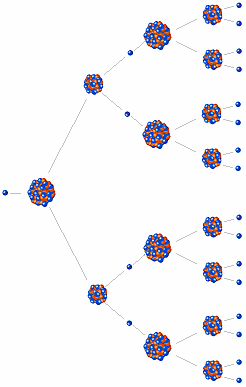
On the reactants' side you have 235 + 1, and on the products' side you have 137 + 97 + ?. You can figure out how many neutrons the reaction will release by balancing the mass numbers. Now you have what you need to write the nuclear equation. To find the atomic number of each isotope that takes part in the reaction, take a look at a periodic table. These neutrons, liberated from the nucleus, are free to induce the fission of other uranium-235 nuclei so that a continued chain reaction of fission events occurs. #color(blue)(A)# - mass number - the number of protons + neutrons in the nucleus The use of water as a neutron moderator helps produce a steady rate of reactor power by slowing the neutrons down that will be absorbed by the U-235 and by. When uranium-235 is struck by a free neutron there is a probability that the nucleus will split into two daughter nuclei, multiple excess neutrons and approximately 200 MeV of energy. #color(red)(Z)# - atomic number - the number of protons in the nucleus So, you know that the uranium-235 nucleus must collide with a neutron, which is a subatomic paticle that has no electric charge and a mass approximately equal to that of a proton, 1. When uranium-235 undergoes a neutron-induced fission reaction, it will split into two smaller nuclei, which, in your case, are tellurium-137, #""^137"Te"#, and zirconium-97, #""^97"Zr"#, releasing neutrons and gamma rays in the process.
